
Each period we’ll see new outer shells, while electrons are added moving to the right.”Ĥ. “This is the periodic table, noble gases stable, halogens and alkali react aggressively. How does the number of protons, or the atomic number, differ from one element to the next in the song?Įach element mentioned has one more proton than the previous element mentioned. The atomic number defines the type of atom, or element, that exists. The first 12 elements are: H, He, Li, Be, B, C, N, O, F, Ne, Na and Mg.Ģ. The periodic table is being read like a book: Elements are listed starting from the top, far left element (hydrogen) across the whole row before going down to the next row far left. What is the pattern you observe for the order in which the elements are presented during the song? List the first 12 elements mentioned in the song. But even without a live demo, students should be able to make observations about the physical properties of the elements and observe trends in reactivity.ĭirections for students: After listening to “The Periodic Table Song,” answer the questions that follow.ġ. If you have the resources and equipment to safely do so, you could demo some of these reactions in the classroom. Reaction (explosion) of alkali metals with water 
Play the following suggested videos or similar video clips and ask students to answer the questions that follow.Ĭhemistry of the group w elements (reactions with water) 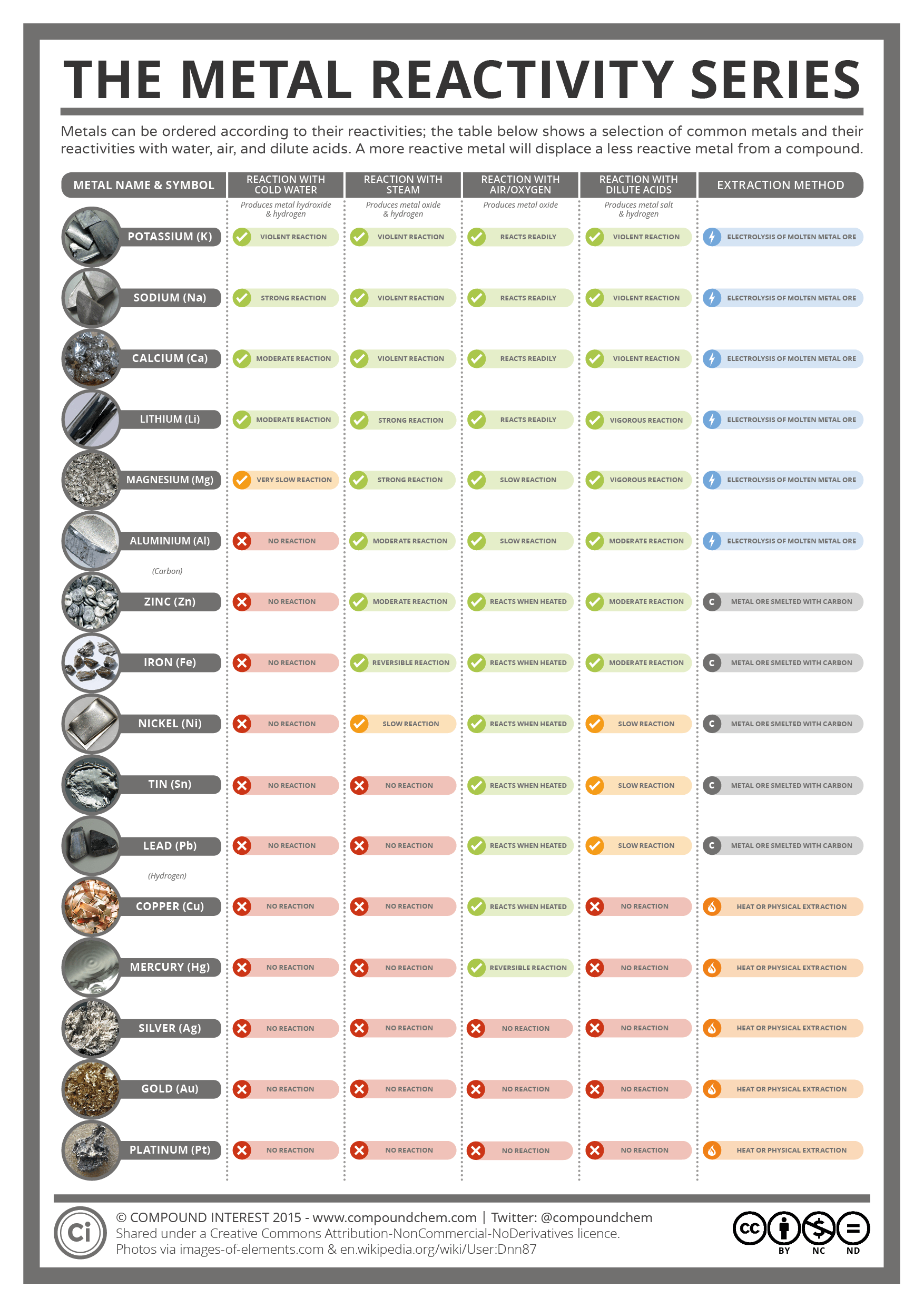
Students should include space to make observations of each nonmetal’s reaction with aluminum. Have students create a second data table to write down the physical properties of each nonmetal in the videos: chlorine (Cl), bromine (Br), iodine (I) and oxygen (O). Students should include space to make observations of each metal’s reaction with water. Have students create a data table to write down the physical properties of each metal element they will see in the video: lithium (Li), sodium (Na), potassium (K), magnesium (Mg), calcium (Ca) and strontium (Sr). After the lesson, you could also share a classic version of the song by Tom Lehrer.Īfter students have completed the first set of questions, they will observe and predict trends in reactivity of metals and nonmetals based on the suggested videos below. Allow students to listen generally once, then ask students to listen again and answer the questions provided. Plan to play the song at least twice to get your students thinking about the table and its general set up. A new version of the song is at Periodic Table Song. 
In the first part of this activity, students will get to know the periodic table through “The Periodic Table Song,” various versions of which are available on YouTube. Handouts showing the periodic table or a large periodic table wall chartĬlassroom computer projector to show video clips demonstrating chemical reactivity A second set of videos focused on reactivity will encourage students to use their observations to identify trends and predict behavior in reactivity among metals and nonmetals. Purpose: After watching a video that introduces them to the periodic table, students will answer questions that prompt them to identify general patterns in the table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
